The atomic number is 4. The atomic mass is 9.012. The number of protons is 4 the number of neutrons is 5. The number of electrons is 4.
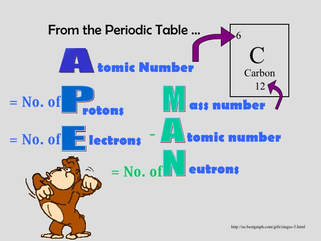
Using A.P.E. M.A.N. A=atomic mass, P=the number of protons, E=the number of electrons
M=Atomic mass A=Atomic number N=number of neutrons, you can find the number of neutrons by subtracting atomic number from atomic mass. So A.P.E.M.A.N. for beryllium would be A=4 P=4 E=4 M=9 A=4 N=5
M=Atomic mass A=Atomic number N=number of neutrons, you can find the number of neutrons by subtracting atomic number from atomic mass. So A.P.E.M.A.N. for beryllium would be A=4 P=4 E=4 M=9 A=4 N=5
